Please click the button below to go to our email login page
|
IF of 12.8! Hongbin Ji’s group cooperated with other teams found the EML4-ALK fusions drive lung adeno-to-squamous transition and its drug resistance through JAK-STAT pathway activationJAK-STAT signaling pathway has been considered as one of the central communication nodes in cell function. How can we design research projects around this pathway?
Today, we’ll share a paper published on J Exp Med with an IF of 12.8, hoping to inspire you from different aspects.
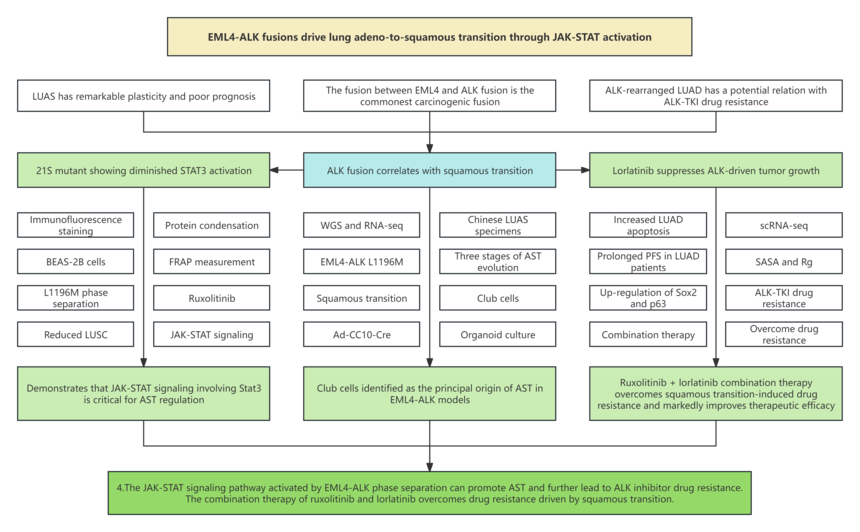
Research background 1. In non-small cell lung cancer, lung adenosquamous carcinoma (LUAS) has mixed pathology of adenoma and squamous cells, which has significant cancer plasticity and poor prognosis. 2. The fusion between echinoderm microtubule-associated protein-like 4 (EML4) and anaplastic lymphoma receptor kinase (ALK) is one of the most prevalent oncogenic fusions in lung cancer. 3. Most ALK-rearranged lung tumors have been pathologically diagnosed as LUAD. The research proved that these tumors exhibit multiple morphologies: solid, acinar, papillary, cribriform, mucin production, and signet-ring components. 4. Several studies indicated that the histological transition of ALK-rearranged LUAD has a potential relation with ALK-TKI drug resistance.
Technical Routes
Research results 1. EML4-ALK drives AST in GEMM 2. Single cell RNA seq (scRNA seq) reveals the evolutionary pathway of AST 3. CC10+cells are the main origin cells for squamous cell transition 4. The organoid system summarizes the AST process 5. EML4-ALK phase separation activates JAK-STAT signaling transduction to regulate AST 6. LUSC shows poor response to TKI treatment 7. LUAD with squamous features has poor response to ALK-TKI 8. The combination therapy of ruxolitinib and lorlatinib results in significant tumor regression Research conclusion The research team utilizes whole genome sequencing (WGS) and RNA sequencing (RNA-seq) of Chinese LUAS specimens. The frequency of ALK fusion is markedly higher adenocarcinoma in LUAS, denoting that ALK fusion may be a potential molecular event driving the adenocarcinoma transition. To further explore its function, researchers constructed an EML4-ALK-driven lung cancer mouse model and analyzed clinical lung cancer samples. Through pathological and single-cell transcriptome analysis, thety found EML4-ALK positive LUAD can transform into LUAS. Cell lineage studies have found that club cells are the main origin cells for EML4-ALK driven transdifferentiation events in LUAS. Moreover, they confirmed that EML4-ALK undergoes phase separation within tumor cells, activating downstream PI3K-AKT, MAPK, and JAK-STAT pathways. Both transformed squamous cell carcinoma and LUAD with gene expression characteristic of squamous cell carcinoma exhibit resistance to ALK inhibitors. Clinical sample analysis confirmed that squamous cell carcinoma patients treated with ALK inhibitors exhibit obviously shortened progression free survival. Re-biopsy samples from recurrent patients also show a significant increase in biological markers of squamous cell carcinoma. Research has shown that the combined use of JAK1/2 inhibitors and ALK inhibitors in mouse models can effectively overcome drug tolerance mediated by LUAS transition. |